Fast Forward to Impact
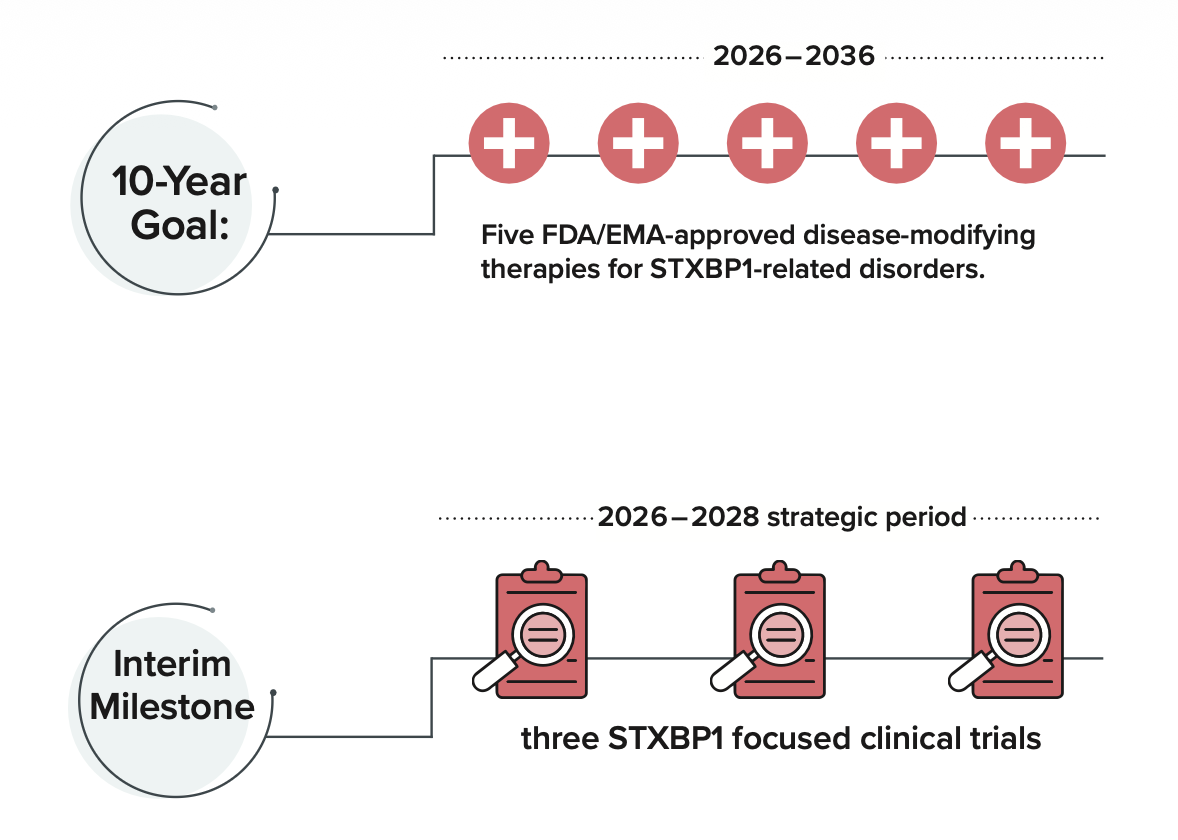
Strategic Plan 2026-2028
Fast Forward to Impact, our Strategic Plan 2026–2028, outlines how the STXBP1 Foundation will accelerate research, strengthen clinical trial readiness, build biopharma interest, and support families today. The plan reflects input from families, researchers, clinicians, industry partners, and Foundation leadership, and is designed to guide our work over the next three years.
The STXBP1 Foundation’s Strategic Plan 2026–2028 builds on the progress of our previous plan and outlines the next phase of our work. Developed with input from families, clinicians, researchers, industry partners, and Foundation leadership, this plan will guide how we accelerate therapeutic progress while continuing to support individuals and families impacted by STXBP1-related disorders today.
For families, this plan means continued investment in resources, education, community connection, and readiness for future clinical trials. For researchers and clinical partners, it means stronger data infrastructure, clearer collaboration pathways, and a Foundation committed to accelerating progress. For biopharma partners, it means a more organized, prepared, and engaged STXBP1 ecosystem.
Why We Believe Progress is Possible
The role of STXBP1 in disease was only discovered in 2008, and the STXBP1 Foundation established in 2017. Despite the newness and rarity of our disorder, research is progressing quickly and we are optimistic that viable therapeutics are possible because of these four important learnings:
The root cause is known - changes in the STXBP1 gene cause STXBP1 disorder
We have a strong understanding of STXBP1’s biological function in the brain
We have a much better understanding of the clinical picture of STXBP1 disorders now than we did even 3 years ago.
Restoring levels of the wildtype STXBP1 protein reverses some disease-relevant symptoms in mice

Priority Areas
Propel STXBP1 - Biotech and Foundation Enabled Development Pathways
Goal 1: Activate Biopharma Partnerships and Capital
Goal 2: Build Trial Readiness Infrastructure
Goal 3: Advance STXBP1 Programs from Discovery to Clinical Readiness.
Support Patients Today
Goal 1: Deliver a High-Quality Annual Summit
Goal 2: Equip Families Through Clear, Transparent, Actionable, and Two-Way Communication
Goal 3: Reduce Barriers to Participation
Prepare for Clinical Trials
Goal 1: Extend and Enhance the STARR Natural History Study
Goal 2: Launch a Family Trial Preparedness Program
Strategic Initiatives
STXBP1 Impact Fund
The Impact Fund will provide a potential mechanism for catalyzing early-stage therapeutics, biomarkers, and enabling technologies, with any financial returns reinvested into STXBP1 research
Global Brand Integration
Strengthen collaboration across the international STXBP1 ecosystem through aligned messaging, shared tools, and voluntary brand coordination — formalized progressively as engagement deepens.